About DME
|
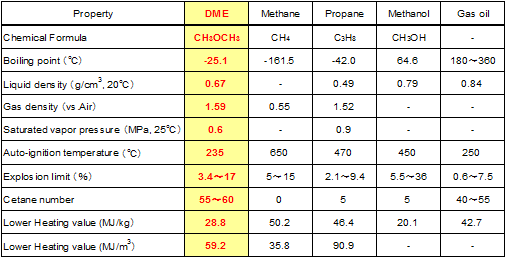
About DME DME (Di-Methyl Ether) is a substance of which the chemical formula is shown by CH3OCH3. DME is a colorless gas at ambient temperature as its boiling point is -25.1℃. As its saturated vapor pressure is as low as 0.6Mpa, it is easily liquefied. As the physical properties of DME are similar to those of propane and butane, main components of Liquid Petroleum Gas (LPG), the storage and handling technology of LPG is applied to DME. As DME has no carbon-carbon bond, no PM (Particulate Matter) is generated on burning. It doesn't contain sulfur not to emit SOX. CO2 emission per calorie is little and NOX emission is little. It is considered "Next generation's clean energy" of which the negative environmental impact is extremely small. Physical properties of DME as fuel are shown in Table 1 in comparison to other related fuels.
Table 1 Physical properties of DME and related fuels
DME can be produced from wide range of raw materials; not only from the fossil resources such as natural gas and coal, but also from the renewable resources such as biomass and hydrogen generated by photovoltaic or wind power electricity. Like LPG and natural gas, DME can be used for wide usage such as home fuel, power generation fuel, diesel engine fuel, and furthermore fuel for the fuel cell. In China where the air pollution is severe, DME is mixed with propane and used for city gas. In United States, DME is expected as diesel alternative with low environmental load and running test of DME diesel truck is going on.
As for quality standardization of DME, JIS K2180 of fuel DME was issued in 2013. In the United States, ASTM D 7901 (Standard Specification for Dimethyl Ether for Fuel Purposes) is issued in 2014. Standardization task of ISO for fuel DME was led by Japan and ISO 16861 (Specifications of dimethyl ether (DME)) is issued in 2015. |
| « prev | top |
 Japanese
Japanese English |
English |